Computational Molecular Biology 2016, Vol.6, No.1, 1-20
6
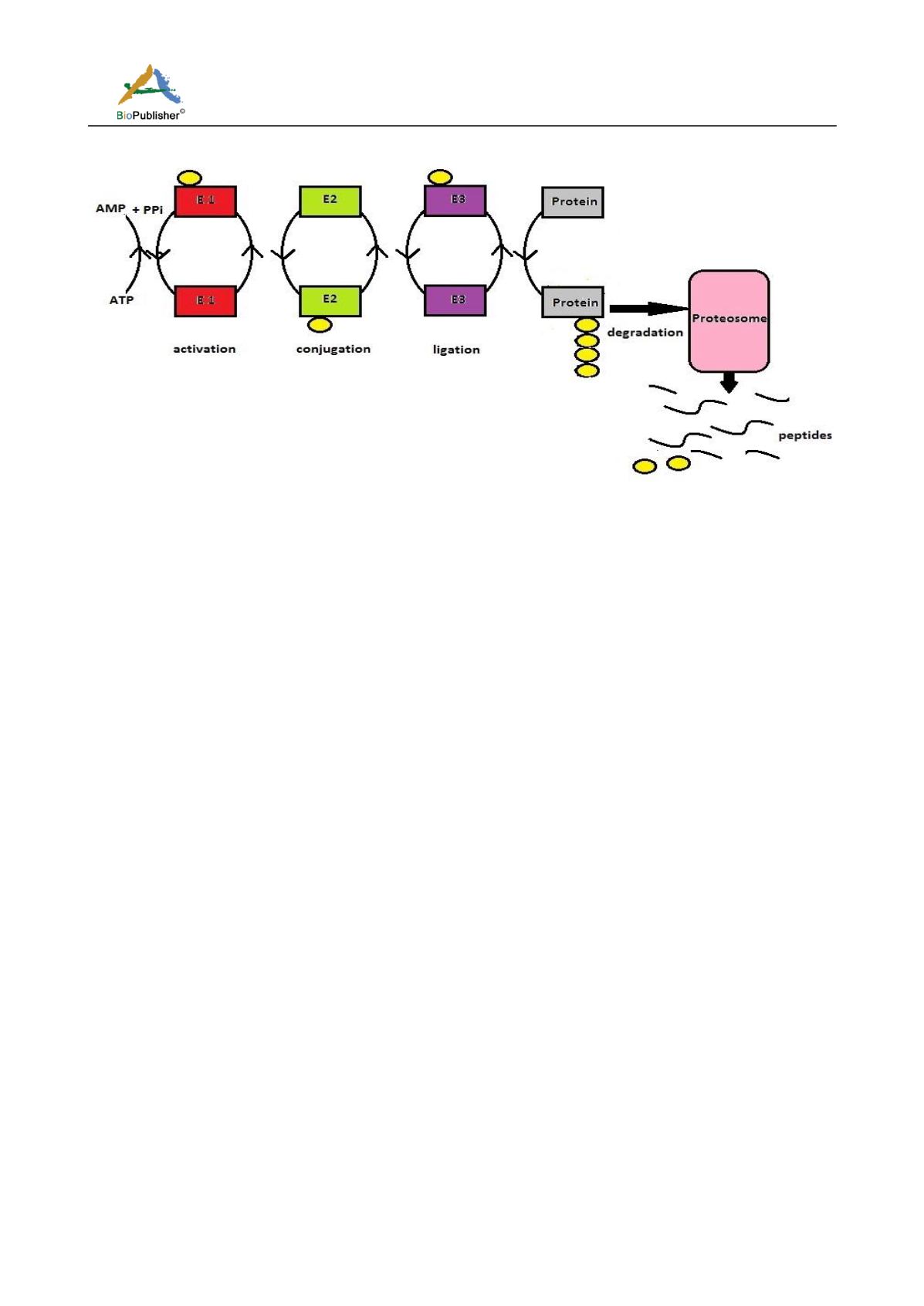
Figure 1 Schematics of UPS pathway
Note: It is a cascade mediated by the interplay of three enzymes, E1(red), E2(green) and E3(purp le). Ubiquitin (yellow) is activated
by binding with E1 (activation) which is then transferred to E2 (conjugation) and lastly to E3 ligase (ligation). E3 transfers the
ubiquitin to the target protein (grey). As the cycle goes on, the protein is poly -ubiquitinated and is finally targeted to the proteosome
(pink) for degradation. Ubiquitin molecule becomes free and is cycled back. It is an energy consuming step.
The E3 ubiquitin ligases are the one which recognize the target proteins and prepare it for proteosomal
degradation. Hence, E3 ligases confer more specificity to the reaction than E2 or DUBs and can be a potential
drug target conferring specific inhibition to the NFkB pathway. Selected E3 ligases are considered good
therapeutic targets as discussed:
4.1.1 HECT
The HECT domain is a bi-lobed structure. It comprises of an N-terminal and a C-terminal lobe tethered by a
flexible hinge. The larger N-terminal lobe contacts with the E2 enzyme whereas the smaller C-terminal lobe
which contains the reactive site Cys residue. The hinge region helps in positioning the active site Cys residue of
E2 with the E3 during ubiquitination. The thioester bond formation requires conformational changes which
involves changes in the orientation of each lobe (Maspero et al., 2013). There are ~30 HECT domain E3s in
mammals. The HECT domain is positioned at the C-terminus of E3 enzymes. The terminal 60 amino acid residues
of the C-terminal lobe contribute to the specificity of ubiquitin chain linkage. The HECT E3s are grouped into
three families : the Nedd4 family, the HERC family and other HECTs (Scheffner et al., 2014).
The Nedd4 HECT E3s consist of nine members. The structure of Nedd4 family E3s have a C2 domain at the
N-terminus, 2-4 WW domains and a HECT domain at the C-terminal end. The WW domain identifies prolinne
rich consensus sequences, PPXYor PY. They also interact with phosphorylated Ser and Thr residues. Some of the
members of Nedd4 along with their cellular functions are: ITCH (cell differentiation and signaling), NEDD4L
(cellular uptake) , SMUR1/2 (cellular signaling) and NEDD4 (transcription)(Ingham et al., 2005; Scheffner et al.,
2014). Many tumor suppressor proteins act as substrate for the HECT E3s, hence revealing their oncogenic
potential. Also, genetic mutation in some of the family members of HECT E3s leads to cancer progression
(Bernassola et al., 2008). Human HERC family consists of six members and is categorized on the basis of
molecular mass, HERCs of more than 500 kDa are placed in one category and HERCs of molecular mass of
~120-130 kDa are placed in other category. HERCs consist of a characteris tic RLD domain. Smaller HERCs carry a
single RLD whereas larger HERCs have more than one RLD. A classical RLD consists of seven-repeats of 50-60
amino acids, it was first demonstrated for RCC1 protein. The activity of the RLD of HERCs is still not clear. HERCs
are believed to be evolved from the nematodes. Other HECTs exhibit domain variations (Hochrainer et al., 2005).


