Basic HTML Version

Molecular Entomology 2013, Vol.4, No.1, 1-5
http://me.sophiapublisher.com
2
plays a crucial role in the Bt function (Baxter et al.
2011).
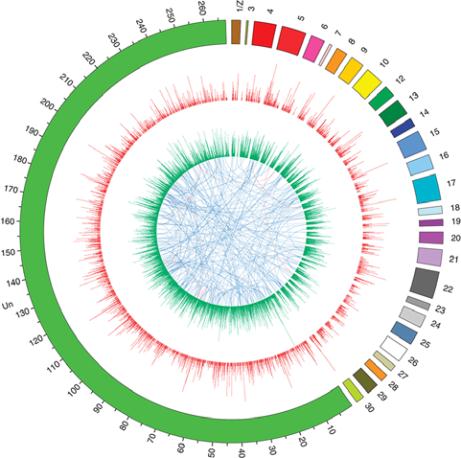
Figure 1 Genomic variations within the sequenced
P. xylostella
strain (adopted from You et al., 2013)
Notes: The outermost circle shows the reference genome
assembly with a 100-kb unit scale. Scaffolds that could be
assigned to linkage groups were joined in arbitrary order to
generate the partial sequences of 28 chromosomes.The green
segment represents the scaffolds that were unable to be
assigned (Un). The innermost circle denotes segmental
duplications (of ≥ 8 kb), with connections shown between
segment origins and duplication locations. Segmental
duplication pairs with 100% similarity are shown in red, and
those with ≥ 90% similarity are shown in blue. Histograms
indicate the number of SNPs (red, outer circle) and indels (light
green, inner circle) in 30-kb and 50-kb windows, respectively
3 Molecular evolution of Diamondback moth
Plutella xylostella
(Linnaeus, 1758) belongs to the
family Plutellidae of Lepidoptera order in the Insecta
class, Arthropoda phylum and Animalia kingdom.
Diamondback moth diverged from two other
lepidopterans
Bombyx mori
and black monarch
butterflies (
D. plexippus
) about 124 million years ago.
Based on the existing insect genomic phylogeny,
diamondback moth was confirmed as a basal and
primitive lepidopteran insect (You et al., 2013).
Phylogenetic analysis further confirmed that the
estimated divergence time of insecta orders was about
265~332 million years ago, which is consistent with
the divergence of monocotyledonous and
dicotyledonous plants 304 million years ago,
indicating the co-evolution of insect behavior and host
plant existence. When the cruciferous plants diverged
from the Caricaceae about 54~90 million years ago,
the diamondback moth also evolved to become a
cruciferous plant-eating insects (You et al., 2013).
4 Molecular adaptation of diamondback moth
The diamondback moth originated in Europe. It was
first found in North America in Illnois in 1854, and it
spread to Florida and the Rocky Mountains in 1883. It
was reported in British Columbia in 1905. Nowadays
diamondback moth can be found in every corner of
the planet where cabbage is planted (Furlong et al.,
2012). Clearly, DMB has evolved to adapt to a variety
of environmental challenges as a result of the complex
network of genes preferentially expressed in the larval
stage that contributes to odorant chemoreception, food
digestion and metabolic detoxification.
DMB has become particularly resistant to chemical
and biological pesticides in recent years. In the 1950s,
Diamond moth was reported to have evolved
resistance to DDT(Ankersmit et al., 1953), followed
by a decline in the effectiveness of pyrethroid
insecticides in the 1980s and reports of Diamond
moth’s resistance to Bt toxins in the 1990s (Heckel et
al., 1999; Tabashnik et al., 1999). Thereafter, virtually
all kinds of pesticides have became ineffective. This
adaption to insecticides contributes to the formation of
biological detoxification pathways in long-term
evolutionary process (You et al., 2013), and the ability
to detoxify many chemical pesticides makes diamond
moth a notorious lepidopteran pest.
References
Ankersmit G.W., 1953, DDT-resistance in
Plutella maculipennis
(Curt.)
(Lep.) in Java, Bull. Entomol. Res., 44(3): 421-425
http://dx.doi.org/10.1017/S0007485300025530
Baxter Simon W., Francisco R. Badenes-Pérez, Anna Morrison, Heiko Vogel,
Neil Crickmore, Wendy Kain, Ping Wang, David G. Heckel, and Chris
D. Jiggins, 2011, Parallel evolution of Bacillus thuringiensis toxin
resistance in Lepidoptera, Genetics, 189: 675-679
http://dx.doi.org/10.1534/genetics.111.130971
PMid:21840855 PMCid:3189815
Baxter S.W., Davey J.W., Johnston J.S., Shelton A.M., Heckel D.G., Jiggins
C.D., and Blaxter M.L., 2011, Linkage Mapping and Comparative
Genomics Using Next-Generation RAD Sequencing of a Non-Model
Organism, PLoS ONE, 6(4): e19315
http://dx.doi.org/10.1371/journal.pone.0019315
Furlong, M.J., Wright, D.J., and Dosdall L.M., 2007, Diamondback moth
ecology and management: problems, progress, and prospects, Annu.
Rev. Entomol., 58: 517-541
http://dx.doi.org/10.1146/annurev-ento-120811-153605
Heckel D.G., Gahan L.J., Liu Y.B., and Tabashnik B.E., 1999, Genetic
mapping of resistance to Bacillus thuringiensis toxins in diamondback
moth using biphasic linkage analysis, Proc. Natl. Acad. Sci. USA.,
96(15): 8373-8377
http://dx.doi.org/10.1073/pnas.96.15.8373
Tabashnik B.E., Huang F.N., Ghimire M.N., Leonard B.R., Siegfried B.D.,

