Basic HTML Version

International Journal of Marine Science 2014, Vol.4, No.1, 1-15
http://ijms.sophiapublisher.com
2
cases in which coral and seagrass co-inhabit the same
area.
Ninomiya et al. (2006) suggested that there is a
physically mutualistic relationship between coral and
seagrass in mixed habitats. The study asserts that the
vertical and horizontal entwining of seagrass stems
and coral branches both on and under the surface of
the seafloor gives stability to seagrass beds. Jompa
and McCook (2003) also showed that in the case of
canopy forming macrophytes, understory corals are
often protected from bleaching damage by shading.
This implies that a dense seagrass community formed
around fragmented coral can encourage coral survival
by providing protection from bleaching.
Manzellos et al. (2012) suggested that coral
calcification rates are higher near seagrass beds,
because seagrasses draw down CO
2
in primary
production and can store the carbon as biomass in
their root systems. Thus, t
here is likely a
biogeochemical relationship between seagrass and
coral. Through the analysis of carbon and inorganic
nitrogen dynamics, this study aims to elucidate this
biogeochemical relationship. In Bise, Okinawa, Japan,
there is a high population density of co-inhabiting
seagrass and corals. Here, the southern portion of this
reef moat is co-inhabited by the coral species,
Montipora digitata
and the seagrass species,
Thalassia
hemprichii
. By comparing the biogeochemical
interactions in the sand, seagrass, coral, and acorn
worm habitats, with those in the coral-seagrass mixed
habitat, we could identify the roles that each
component has in the carbon and inorganic nitrogen
dynamics both separately and interactively within the
coral reef ecosystem.
2 Materials and Methods
2.1 Study site
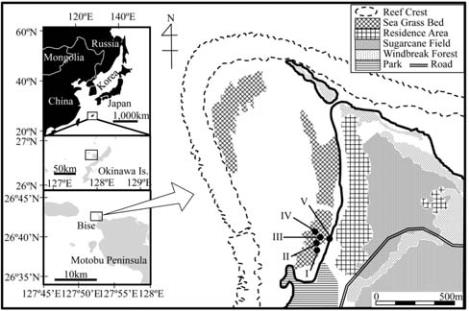
This study was conducted in the subtropical reef moat
off of Bise Coast, Okinawa, Japan, from Aug. 1 to 6,
2008 (Figure 1). It is characterized by coral, seagrass,
and coral-seagrass mixed communities. The dominant
coral species is
Montipora digitata,
and the dominant
seagrass species is
Thalassia hemprichii
. The most
conspicuous benthic organisms inhabiting this area are
the acorn worms;
Schizocardium
sp. and
Ptychodera
flava
. These invertebrates are bioturbators which
produce fecal casts on the sediment surface while
burrowing into the coral reef sands (up to 24
indivduals m
-2
, pers. obsv.). The underlying substrate
of these habitats is primarily dominated by limestone.
Figure 1 Map of the study site which shows the sand (I),
seagrass (II), coral-seagrass (III), coral (IV) and sand-acorn
worm (V) habitat
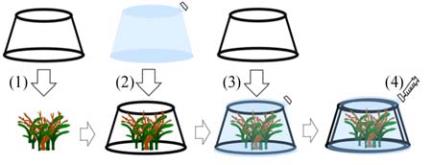
2.2 Chamber experiment
Benthic chambers (Ishikawa et al., 2007) were used to
conduct 2 h incubations in each habitat (Figure 2).
Using gardening stands fitted with plastic bags as
incubation chambers, the nutrient and carbon
dynamics of five benthic habitats were assessed: sand
only (SD, 100% sand), seagrass only (SG, 100%
T.
hemprichii
), coral-seagrass (CS,
T. hemprichii
: 50%,
M. digitata
: 45%, sand : 5%), coral only (CR,
M.
digitata
: 95%, sand : 5%), and acorn worm habitats
(AC, 100% sand with 2-4 acorn worms). Three
incubation chambers were placed in each habitat and
water samples were collected from each chamber. For
the SG, CS, and CR habitats, 44 L benthic chambers
(1479 cm
2
base area, 40 cm ht.) were deployed.
Smaller chambers were used for the SD and AC
habitats, 15 L benthic chambers (984 cm
2
base areas,
Figure 2 Schematic diagram of the benthic chamber. First, hard
flames are put on each habitat (1). Then, clear plastic bags were
covered on the flames (2). And then, hard flames was put again
on the plastic bags (3). Water samples are taken by using
syringes before and after the 2-h chamber incubations (4)

